|
|
| Physician Perceptions of Drug-Drug Interactions and How To Avoid Them |
|
|
SHELDON H. PRESKORN, MD
BERYL SILKEY, ScM
TERESA L. JONES, MPH, MT(ASCP)
|
|
Journal of Psychiatric Practice, March 2002, 112-115
|
Several earlier columns were devoted to case reports illustrating the many varied ways in which drug-drug interactions (DDIs) can present.1 These case reports document how DDIs can be a cause of significant patient morbidity and increased health care costs. In the last column,2 the sheer magnitude of the number of potential drug combinations was discussed. Since that column was written, the authors made a grand rounds presentation at a Veterans Administration (VA) Medical Center to a group composed mainly of primary care physicians. There were 35 participants: 16 faculty, 12 residents, and 7 medical students. Of 21 participants who completed a questionnaire, 11 were VA staff, 9 were non-VA, and the affiliation of 1 was not listed. The primary focus of the presentation was to review the findings from a study of the nature and frequency of polypharmacy in the VA. The presentation employed a Power Poll technology that allowed the audience to anonymously respond to specific queries built into the presentation. These queries elicited the opinions and knowledge of the audience about various issues related to DDIs. This column will review those responses.
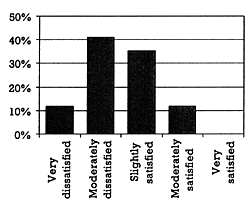
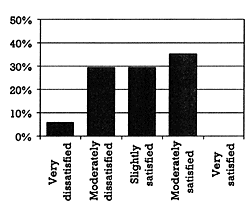
| Figure 1 - How satisfied are you with your ability to understand and avoid drug/drug interactions? | a. Pre-Presentation
b. Post-presentation
 | | Figure 2 - What proportion of patients at your VA are on a unique combination of drugs? | 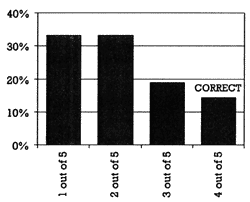 | | Figure 3 - Which of the following drugs is not a substantial CYP2D6 inhibitor? | 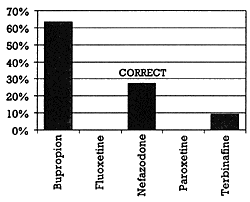 | | Figure 4 -What percent of the 44 cases are on a dose that might lead to serious toxicity? | 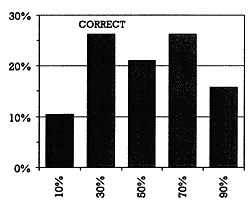 | | Figure 5 -What percent of the 43 pairs are listed in the VA drug alert system? | 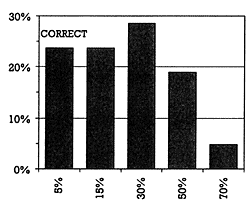 |
The presentation began with a question about how satisfied the individual prescriber was with his or her ability to understand and avoid DDIs (Figure 1a). This question was repeated after the data had been presented and discussed (Figure 1b). At that time, there was a noticeable shift to the left (less satisfied): Over 50% of the audience reported being either "moderately" or "very dissatisfied" with their ability to understand and avoid DDIs; 10% reported being "very dissatisfied," which was double the percentage before the formal presentation began.
Although the audience tended to overestimate the average number of drugs that an average VA patient is taking, they substantially underestimated the number of patients who were on a unique combination. Only 12% correctly chose 4 out of 5 (80%) as the proportion of A patients on a unique combination of medications (Figure 2).
The inhibition of drug metabolizing enzymes is perhaps the most common mechanism underlying clinically important pharmacokinetically mediated drug-drug interactions.3 Cytochrome P450 (CYP) enzymes are responsible for the bulk of phase 1 drug metabolism. For this reason, the inhibition of these enzymes is a clinically important issue and has led to the removal of some drugs from the market.2 Two of these enzymes, CYP 3A and CYP 2D6, are responsible for approximately 50% and 30% of oxidative drug metabolism, respectively. For this reason, the audience was asked which of the following drugs is not a substantial cytochrome P450 (CYP) 2D6 inhibitor: bupropion, fluoxetine, nefazodone, paroxetine, or terbinafine (Figure 3). Only 25% correctly identified nefazodone. The majority (60%) misidentified bupropion as a drug that does not substantially inhibit CYP 2D6.
CYP 2D6 is the rate-limiting enzyme for the metabolism of all of the tricyclic antidepressants (TCAs). That is clinically important because TCAs have a narrow therapeutic index so that concentrations that are only four to five times higher than those needed to treat clinical depression can cause serious toxicity.4 Coadministration of a substantial CYP 2D6 inhibitor, such as bupropion, fluoxetine, paroxetine, or terbinafine, can reduce the clearance of TCAs to the extent that the routine doses needed for antidepressant efficacy can produce potentially toxic levels.
In the VA polypharmacy study, 44 cases were identified in which a TCA was coprescribed with a substantial CYP 2D6 inhibitor. The audience was asked what percentage of these patients they believed were on a TCA dose that could produce potentially toxic levels in the presence of substantial CYP 2D6 inhibition. The correct answer was 30% of the 44 cases. In other words, one out of approximately every three patients in the VA on a TCA and a coadministered substantial CYP 2D6 inhibitor was at risk for serious TCA-induced toxicity. While that number is high, 6 out of 10 respondents anticipated that even more patients would be at risk (Figure 4). These facts underscore the clinical importance of this information.
To extend this approach, the VA database was examined for the occurrence of potentially dangerous drug pairs based on the mechanism of CYP enzyme inhibition. Two types of drugs were identified: a) those that were known to be substantial inhibitors of specific CYP enzymes, and b) those that were principally metabolized by a single CYP enzyme and for which a significant reduction in clearance would produce a clinically important change in the dose-response curve (e.g., TCAs). The co-occurrence of these two types of drugs were then examined in the VA database, and 43 different potentially dangerous drug pairs of this type were identified. The audience was then asked how many of these potentially dangerous pairs were in the VA drug alert system as it existed at the time of the study. 80% of the audience thought 15% or more of these potentially adverse drug combinations would have been identified by the alert system. In fact, only 5% were (Figure 5).
The reason so few combinations were listed was that, at the time that the study was conducted, the VA drug alert system listed only drug combinations for which there were published studies demonstrating the risk of a potentially serious DDI. The alert system did not identify the potential for a serious adverse DDI if there were no published studies, even if one drug was known to be a substantial CYP enzyme inhibitor and the other drug was known to have a narrow therapeutic index and to be preferentially metabolized by the inhibited CYP enzyme.
At the conclusion of the presentation, the audience was asked to rank the importance of four different ways of reducing the occurrence of untoward DDIs. Increased prescriber education was rated as very important by 65% of the audience, followed by enhancing the drug alert system (60%), greater patient education (50%), and finally revising the formulary (35%).
Despite these ratings, revising the formulary may be the simplest and most effective way of reducing the risk of untoward DDIs. This approach gives preferential status to the drugs within a therapeutic class that have a lower risk of DDIs. For example, this approach would give citalopram or sertraline preferential status over fluoxetine, fluvoxamine, or paroxetine on the basis that citalopram and sertraline, in contrast to the other three SSRIs, do not substantially inhibit any CYP enzymes and therefore do not have the potential to cause CYP-mediated DDIs.
Conclusion
The feedback received from this audience illustrates that prescribers initially underestimate the risk of CYP-enzyme-mediated DDIs and the complexity of the problem. It also demonstrates that physicians are aware of the problems inherent in the practice of polypharmacy. Their dissatisfaction with their ability to understand and avoid DDIs is realistic given the sheer number of potential drug combinations that can occur in clinical practice.2
As discussed in the last column, there are over 3200 prescription drugs listed in the 2002 Physician Desk Reference. This results in over 2.8 x 1015 potential 5-drug combinations. The first new drug approved in 2002 will increase that number by 4.4 trillion. On average, 41 new drugs were approved each year in the 1990s, with each addition further increasing the number of potential drug combinations.
Obviously, no individual can memorize all of the potentially dangerous drug combinations. In fact, most adverse combinations are identified only when they present in an acute and fulminant manner and in sufficient numbers to be detected by the current informal drug surveillance system used in this and most other industrialized countries. Such identification then often leads to studies aimed at identifying the mechanism underlying the DDI. Before a drug is marketed, the number of formal DDI studies done rarely exceeds 10 and these studies look only at the use of the new drug in combination with one other drug. Until recently, it was rare for studies to be done examining the effect of more than two drugs in combination. Although a few studies are now being done examining the effect of more complicated combinations, such as the co-administration of substantial CYP 2D6 and CYP 3A inhibitors with a drug that is a substrate for both of these CYP enzymes, it will be some time before the results of those studies begin to provide sufficient data to meaningfully change drug prescribing practices.
The results of the VA polypharmacy study and the feedback from this survey of an audience of physicians also illustrate the dangers inherent in slavishly relying on current drug alert systems. While such systems can be helpful, they are by no means foolproof. They are constantly being improved and updated. Nevertheless, their greatest limitation is the lack of knowledge about the potential mechanisms -- both pharmacodynamic and pharmacokinetic-mediating DDIs and the enormous number of potential complex drug combinations.
For these reasons, the well trained, astute, careful, and conscientious prescribes continues to play a vital role in the avoidance of adverse DDIs and also in the detection of previously unknown adverse DDIs. In fact, the current knowledge of CYP-enzyme-mediated DDIs was the result of the observations of such clinicians.6
Basic studies were done to follow-up on those clinical observations, which led to the identification of the underlying mechanisms. That knowledge was in turn incorporated into the drug discovery process to minimize the chance of inadvertently developing new drugs with the potential for causing such interactions.5 This pattern of clinical identification of adverse drug combinations followed by basic science studies will likely be repeated over and over in the future as new drugs with new mechanisms are developed.
This issue is particularly important in psychiatry given the sheer number of new targets for drug discovery being identified by the Human Genome Project.7 While pharmacokinetically mediated DDIs were used as the example in this column, the greater challenge in the future for psychiatry will likely be pharmacodynamically mediated DDIs, as an increasing number of drugs with novel central mechanisms of action are used in combination.
References
- Preskorn SH, The polypharmacy section. (collection of columns published in the Journal of Psychiatric Practice). Accessed March 8, 2002
- Preskorn SH, Drug approvals and withdrawals over the last 60 years. Journal of Psychiatric Practice 2002;7:410
- Levy RH, Thummel KE, Trager WF, Hansten PD, Eichelbaum M, eds, Metabolic drug interactions. Philadelphia: Uppincott, Williams & Wilkins, 2000
- Preskorn SH, Outpatient management of depression: A guide for the primary-care practitioner, 2nd Edition. Caddo, OK: Professional Communications; 1999
- Preskorn SH, The human genome project and modern drug development in psychiatry. Journal of Psychiatric Practice 2000;6:272-276
- Vaughan DA, Interaction of fluoxetine with tricyclic antidepressants. Am J Psychiatry 1988;145:1478
- Preskorn SH, Modern drug development and the Human Genome Project. (collection of 4 columns published in the Journal of Psychiatric Practice). Accessed June 25, 2001
|

